CARBON AND ITS COMPOUND
COMPOUND: Two or more elements combine to form
compound.
There
are two types of compounds- Organic Compound and Inorganic
Compounds. Organic compounds are the one which are
made up of carbon and hydrogen.
COVALENT BOND
The bond
formed by sharing a pair of electrons between two atoms are known as Covalent Bond. Carbon forms covalent bond. Carbon exists in two forms- as
free state and as combined state. Free form of carbon is found in graphite,
diamond and fullerene. In combined state, carbon exists as Carbon-dioxide,
Glucose, Sugar etc.
Allotropes of Carbon
Different
forms of an element that has same chemical properties but different physical
properties are known as Allotropes.
There are three allotropes of carbon- diamond, graphite and fullerene.
Diamond
Diamond
exits as three-dimensional network with strong carbon-carbon covalent bonds.
Diamond is hard in nature with high melting point. It shines in presence of
light and it is a bad conductor of electricity. The most common use of diamond
is in making jewellery. It is also used in cutting and drilling tools.
Graphite
Graphite
is made from weak van der wall forces. Each carbon atom is bonded with other
three carbon atoms in order to form hexagonal rings. It serves as good conductor
of heat and electricity. It is used as dry lubricant for machine parts as well
as it is used in lead pencils.
Fullerene
It is a
hollow cage which exits in the form of sphere. Its structure is similar to
fullerene. But along with hexagonal rings, sometimes pentagonal or heptagonal
rings are also present.

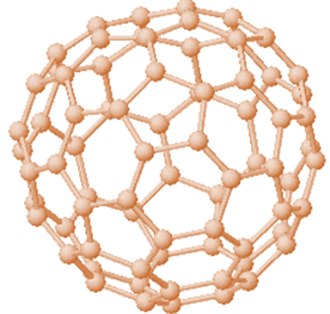
Fig.1 Structure of fullerene
Two Important Properties of Carbon
Catenation
and tetravalency are the two important properties of carbon. Catenation is a
property of carbon by which carbon atoms can link one another via covalent bond
and can form long chains, closed ring or branched chains etc. Carbon atoms can
be linked by single, double or triple bonds. Carbon has a valency of 4 due to
which it is known to have tetravalency. Due to this one carbon atom can bond
with other 4 carbon atoms, with other atoms also such as Oxygen, Nitrogen etc.
Hydrocarbons
Compounds
which are made up of carbon and hydrogen they are known as Hydrocarbons. There are two types of
hydrocarbons found - Saturated Hydrocarbons and Unsaturated Hydrocarbons. Saturated Hydrocarbons
consist of single bonds between the carbon atoms. For Example,
Alkanes. Alkanes are saturated hydrocarbons represented by a formula, CnH2n+2.
Unsaturated
Hydrocarbons are the one with double or triple bonds between the carbon
atoms. For Example, Alkenes
and Alkynes. Alkenes are represented as CnH2n whereas
alkynes are represented as CnH2n-2. Some saturated
hydrocarbons and unsaturated hydrocarbons are represented as -

Fig.2. Saturated hydrocarbons
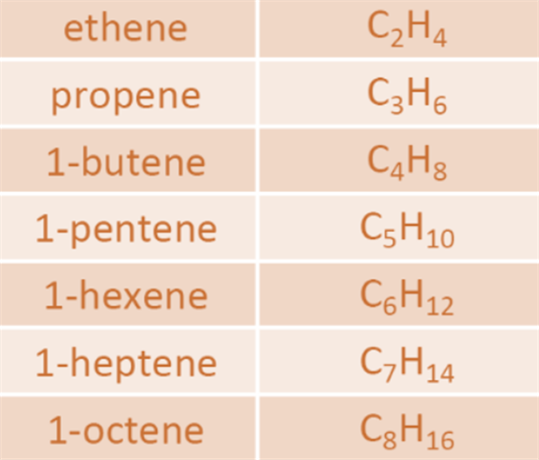
Fig. 3. Unsaturated hydrocarbons
Structure
of hydrocarbons can be represented in the form of electron dot structure as
well as open structures as shown below-
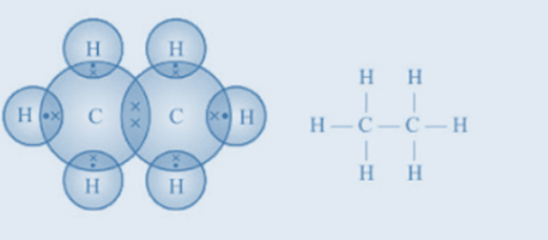
Fig.4. Electron dot structure and open structure of
ethane
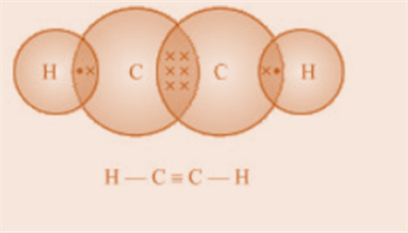
Fig.5. Electron dot structure and open structure of
ethyne
Carbons Compounds based on the basis of structure
Carbon
Compounds can be classified as straight chain compounds, branched chain
compounds and cyclic compounds.They are represented as -
![]()
Fig.6. Straight chain carbon compound
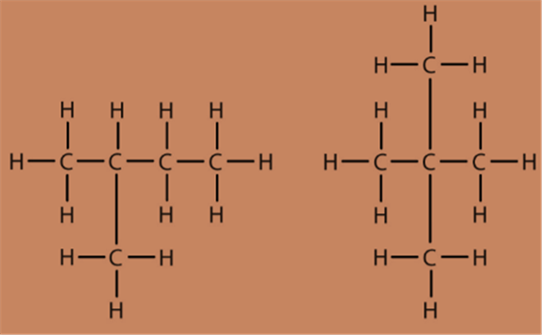
Fig.7. Branched chain compounds
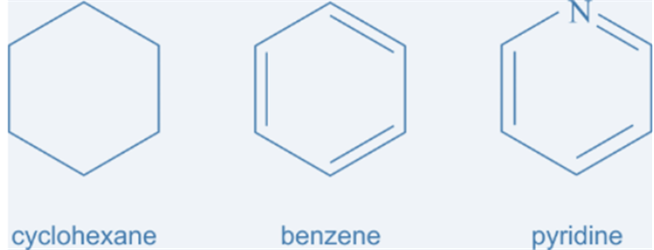
Fig.8. Cyclic carbon compounds
Functional Groups
One of
the hydrogen atoms in hydrocarbon can be replaced by other atoms according to
their valencies. The atoms which decides the properties of the carbon atoms,
are known as Functional Groups. For Example, Cl, Br, -OH, Aldehyde,
Ketone, Carboxylic Acid etc.
Homologous Series
Series of
compounds in which same functional group substitutes for the hydrogen atom in a
chain of carbon.
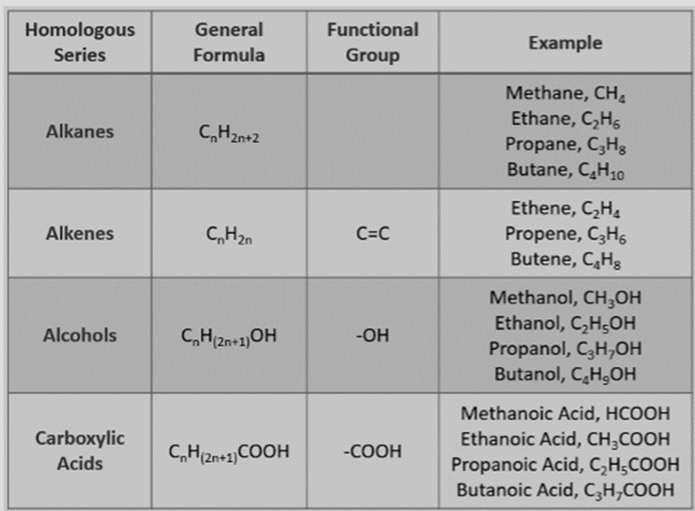
Fig.9. Homologous series
Nomenclature of Carbon Compounds
·
First of
all, identify the number of carbon atoms in compounds. And in it identify the
longest chain
·
Then
functional group can be indicated by suffix or prefix.
·
Cyclic
hydrocarbon is designated by prefix cyclo.
·
If there
are two or more different substituents they are listed in alphabetical order
·
If the
same substituent occurs more than once, the location of each point on which the
substituent occurs is given
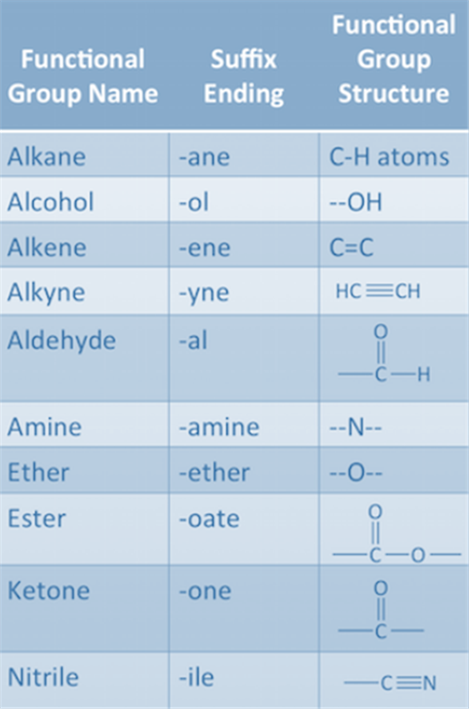
Fig.10. Different functional groups
Chemical Properties of Carbon Compounds
Combustion
Carbon
along with its compound is used as a fuel as it burns in presence of oxygen to
release energy. Saturated hydrocarbons produce blue and non-sooty flame whereas
unsaturated hydrocarbons produce yellow sooty flame.
CH4 + 2O2 → CO2 +
2H2O
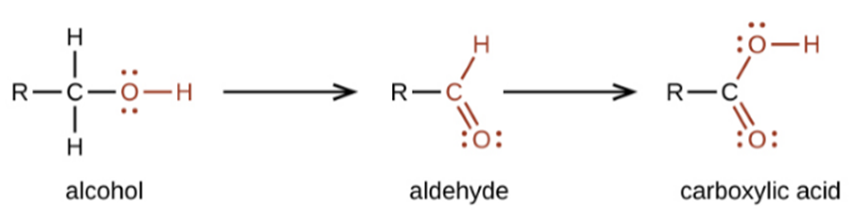
Oxidation
Alcohol
can be oxidized to aldehydes whereas aldehydes in turn can be oxidized to
carboxylic acid. Oxidizing agent such as potassium permanganate can be used for
oxidation.
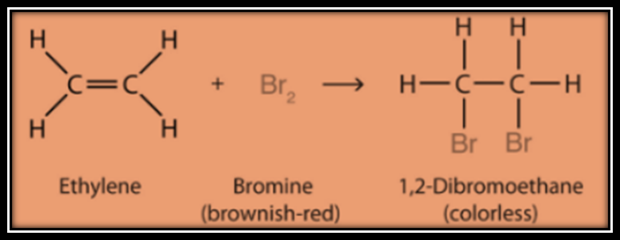
Addition Reaction
Hydrogenation
of vegetable oil is an example of addition reaction. Addition of hydrogen in
presence of catalyst such as nickel or palladium. This converts oil into ghee.
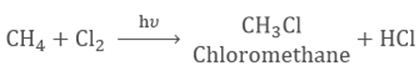
Substitution Reaction
When one
atom in hydrocarbon is replaced by chlorine, bromine, etc. this is known
as Substitution Reaction.
Important Carbon Compounds: Ethanol and Ethanoic
Acid
Ethanol
is a volatile liquid with low melting point. It reacts with sodium to form
sodium ethoxide.
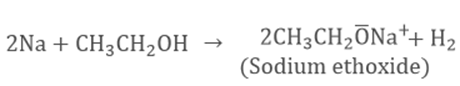
This
above reaction is used to test the presence of ethanol by the evolution of
hydrogen gas.
Dehydration
of ethanol in presence of hot sulphuric acid forms alkene.
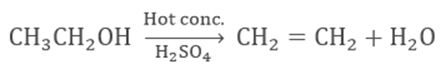
Ethanoic
acid is a colourless liquid. When pure ethanoic acid freeze like ice, it is
known as Glacial Acetic Acid.
It is formed at a temperature of about 16.6 degree centigrade
Ethanoic
Acid/Acetic acid when reacts with ethanol it forms an ester. Ester can be
identified by its sweet smell.
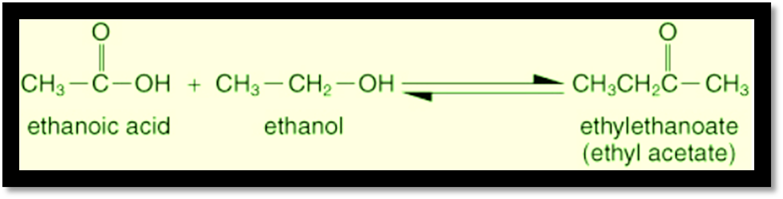
Reaction
of ester with strong base is used to form soap. This is known as Saponification. Acetic acid also
reacts with strong base to form sodium acetate and water.
NaOH + CH3COOH + CH3COONa + H2O
Soaps and Detergents
Sodium or
potassium salt of carboxylic acid is known as Soap. They work most effectively in soap water. Detergents are
sulphonate or ammonium salt of long chain of carboxylic acid. They can work
effectively on soft as well as hard water.
Cleansing Action of Soaps and Detergents
Cleansing
action of soaps and detergents is due to ability to minimize the surface
tension of water, to emulsify oil or grease and to hold them in a suspension of
water. When soap dissolves in water, it forms soap anions and soap cations. The
hydrophobic part of soaps and detergents are soluble in grease and hydrophilic
part is soluble in water.
Soap and Micelle Formation
When dirt
and grease are mixed with soap water, soap molecules arrange them in tiny
clusters known as Micelle.
The hydrophilic part sticks to the water and form outer surface of the micelle
and hydrophobic part binds to oil and grease.